ASH Annual Meeting 2022
Advertisement
The phase II study results are in. Researchers will now test rusfertide against placebo in a randomized clinical trial.
Researchers determined the incidence of TEAEs related to rusfertide in patients with PV.
A new method of peripheral blood stem cell hematopoietic cell transplantation led to a 1-year chronic GVHD rate of 11%.
Patients who received a triplet regimen as GVHD prophylaxis after HSCT had favorable outcomes.
GVHD was only reported in one patient who received veto cells plus haploidentical T-cell-depleted HSCT.
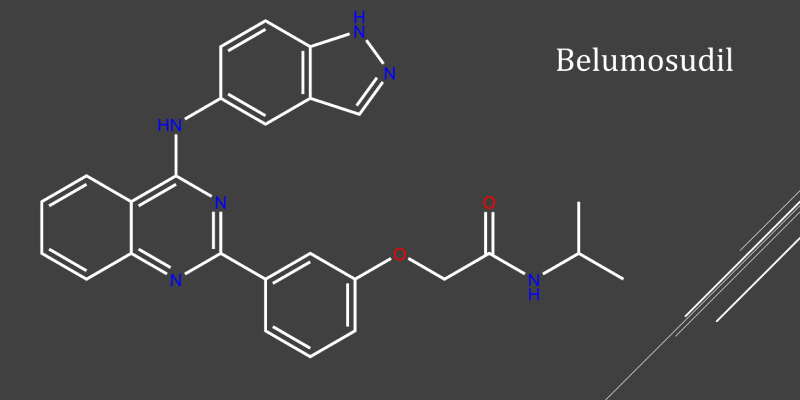
Belumosudil can impact the pharmacokinetics of tacrolimus and sirolimus, which is important for dosing considerations.
Donor age and type can significantly affect the outcomes of patients who undergo hematopoietic cell transplantation.
GVHD prophylaxis with posttransplant cyclophosphamide plus tacrolimus was better than tacrolimus alone for allogeneic HCT.
No GVHD was reported in patients with CD30-positive lymphoma who received CD30.CAR EBVSTs.
GVHD prophylaxis with itacitinib plus tacrolimus/sirolimus was safe and well-tolerated after transplant.
No graft-versus-host disease (GVHD) occurred in patients with multiple myeloma (MM) who received allogeneic CAR-T therapy.
Around 25% of patients with high-risk MDS or AML who received venetoclax plus azacitidine maintenance developed chronic GVHD.
Frailty is strongly associated with worse survival outcomes in patients with chronic graft-versus-host disease (GVHD).
No GVHD exacerbations occurred with a BCMA-directed CAR-T in patients with multiple myeloma.
Donor-derived, CD7-directed, CAR T-cell therapy had “encouraging activity” but led to reversible GVHD in 40% of patients.
Around one-third of patients with TP53-mutated acute leukemias or MDS who underwent HSCT developed chronic GVHD.
No moderate-to-severe chronic GVHD occurred in patients with high-risk hematologic malignancies who received Orca-Q.
Fludarabine plus busulfan led to lower rates of grade III-IV acute GVHD than cyclophosphamide plus busulfan.
Belumosudil combination therapy in a real-world setting “appears safe, tolerable, and effective” for chronic GVHD.
Patients receiving allogeneic HSCT face an average of $1 million in transplant-related costs throughout their lifetimes.









 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.